
Laboratory Investigations in Microbiology

 |
Laboratory Investigations in Microbiology |
 |
Culture media
Many bacteria can be cultured, or grown, in the laboratory by making use of
specific culture
 media. Culture media contain all the nutrients necessary to
support the growth of the desired microbes. Culture media whose ingredients
are known exactly are called simple, or defined, media. Culture media whose
composition is not exactly known because they contain ground-up plant or
animal tissue are called complex media. Complex media support a wide variety
of microbes and are better suited as all-purpose media. We will most
frequently use a complex medium known as Trypticase Soy (agar or broth) in
this lab.
media. Culture media contain all the nutrients necessary to
support the growth of the desired microbes. Culture media whose ingredients
are known exactly are called simple, or defined, media. Culture media whose
composition is not exactly known because they contain ground-up plant or
animal tissue are called complex media. Complex media support a wide variety
of microbes and are better suited as all-purpose media. We will most
frequently use a complex medium known as Trypticase Soy (agar or broth) in
this lab.
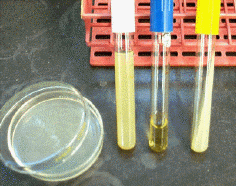
Culture media that are liquid are known as broths (C). With the addition of agar (a polysaccharide made by some algae), culture media can be made to solidify. When heated to 80°C, agar solutions are liquid, but will gel when cooled to below 40°C. Liquid agar media can be poured into Petri dishes or test tubes to harden there. In this way, one can produce agar plates (A), agar slants (D; agar hardens in a test tube at an angle), and agar deeps (B; the tube is mostly filled with solidified agar). Agar plates are usually used for isolating pure cultures of bacteria and for observing colony characteristics. However, the large surface area makes them more susceptible to contamination and to drying out. Agar slants are an excellent means for keeping a pure culture for several months. A smaller surface area and a much smaller opening keep moisture in and contaminants out. For longer-term cultures, agar deeps can be used. Deeps are stab-inoculated with an inoculating needle (see Ch. 3). Bacteria can last months to a year or more. Broths, in contrast, allow for very rapid growth of bacteria, but nutrients are quickly depleted and wastes accumulate. Broths are most often used to grow up large numbers of bacteria quickly.
At times it is desirable to restrict the growth of some bacteria while allowing others to grow successfully. Culture media that accomplish this are termed selective media. Examples include Eosin Methylene Blue agar (EMB), which favors Gram-negative bacteria, and Mannitol Salt Agar (MSA), which favors Staphylococci. At other times, we want to distinguish between similar-looking bacteria based on biochemical abilities such as lactose fermentation. The use of a differential medium such as blood agar allows us to do this. EMB and MSA also happen to be differential. Bacteria which can ferment lactose have purple/green colonies on EMB, while those that cannot have pink/white colonies. On MSA, bacteria that can use mannitol change the agar to yellow; those that do not use mannitol leave the agar unchanged (or may even turn it red/magenta).
Table 1. Types of complex culture media used in Microbiology
| Medium | Selective | Differential | Enriched |
| Trypticase soy agar/broth | NO | NO | NO |
| Nutrient agar/broth | NO | NO | NO |
| citrate agar | NO | YES (citrate utilization) | NO |
| SIM agar | NO | YES (H2S and indole production; motility) | NO |
| Motility agar | NO | YES (motility) | NO |
| Eosin Methylene blue agar | YES (Gram negative) | YES (lactose fermentation) | NO |
| Mannitol salt agar | YES (Gram positive) | YES (mannitol fermentation) | NO |
| Brilliant Green Lactose Bile broth | YES (Gram negative) | YES (lactose fermentation) | NO |
| Phenol red (dextrose/lactose/sucrose) broth | NO | YES (glucose/lactose/sucrose fermentation) | NO |
| MR-VP medium | NO | YES (butanediol or mixed acid fermentation) | NO |
| Violet Red Bile agar | YES (Gram negative) | YES (lactose fermentation) | NO |
| Lactose broth | NO | YES (lactose fermentation) | NO |
| Blood agar | NO | YES (hemolysis) | YES |
| Chocolate agar | NO | NO | YES |
Culture media may contain a wide range of ingredients, ranging from soy meal to blood. Often they contain a specific source of protein (tryptone, peptone, casein) which may be enzymatically treated (e.g. "trypticase") to make it smaller/more digestible. Media may also include salts, dyes, pH indicators, and inhibitors such as bile.
Next lab period
© 2003 - 2025 José de Ondarza, Ph.D.